Is stool testing worth it?
What the Science Actually Says About Gut Microbiome Testing—and What to Do Instead
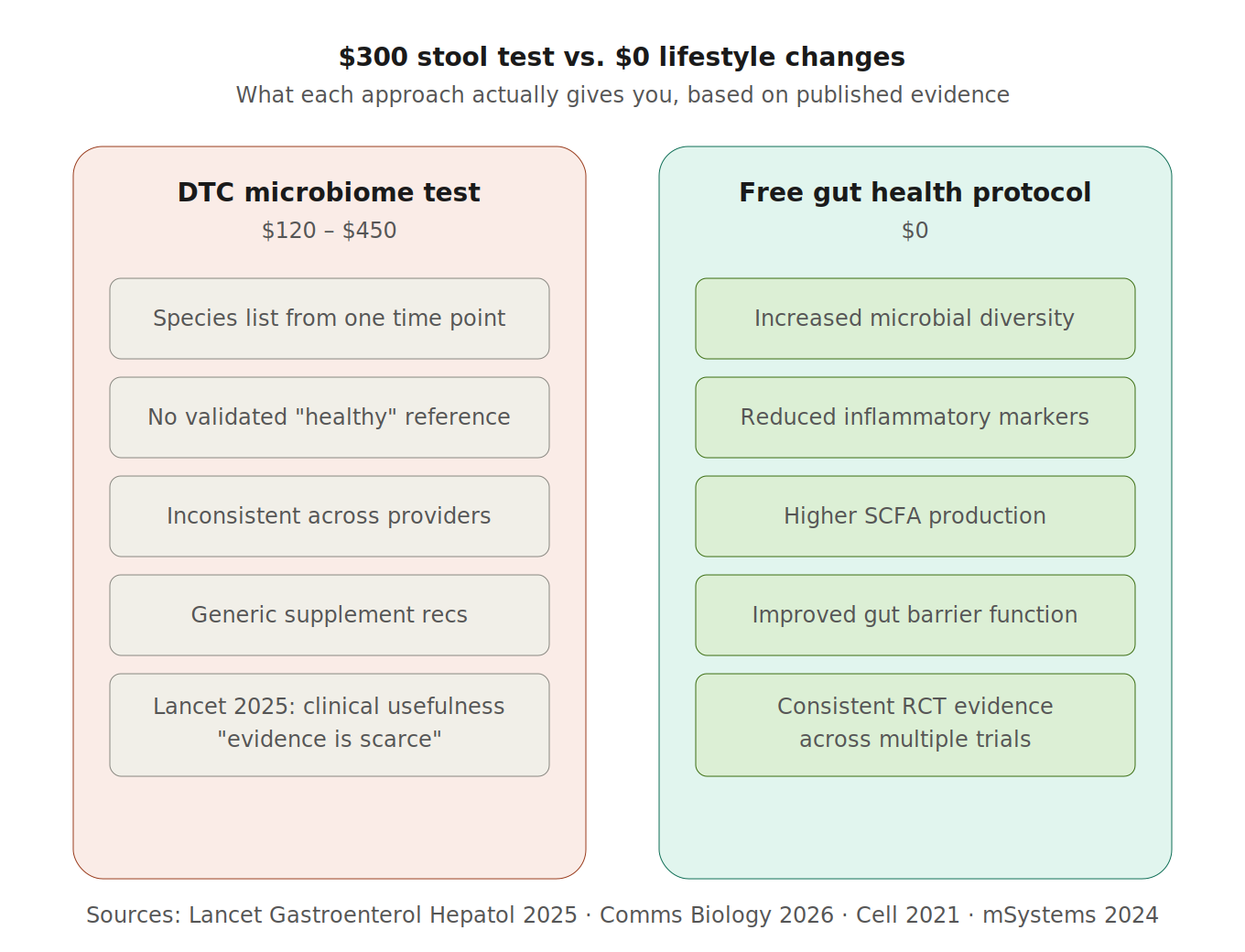
Here’s a scenario I see often. A patient walks in with a 20-page report from a direct-to-consumer gut microbiome test. Glossy charts. Color-coded bacteria. A long list of supplements they’re now convinced they need. They spent somewhere between $200 and $400 for this report, and they want me to tell them what it means.
The honest answer? Usually, not much.
And I say that not because the microbiome doesn’t matter—it absolutely does. It’s one of the most important frontiers in medicine right now. I say it because a lot of the testing, as it exists today, has some serious problems that most consumers don’t hear about. And the things you can actually do to improve your gut health are largely free.
Let’s walk through this.
The Problem With Consumer Microbiome Testing
First, some context. These at-home kits—Viome, Ombre, BIOHM, Zoe, GI-MAP, Gut Zoomer and others—generally run between $120 and $450. You collect a stool sample, mail it in, and get back a report telling you which bacteria live in your gut, how “diverse” your microbiome is, and a list of food and supplement recommendations. The presentation is slick. The marketing is confident. And I understand why people are drawn to them (I’ve been sucked in and tried some of these).
But here’s what the research actually tells us.
In February 2025, a major international consensus statement was published in The Lancet Gastroenterology & Hepatology by a panel of leading microbiome researchers. The conclusion was blunt: evidence supporting the clinical usefulness of these tests is scarce. The panel actually discouraged patients from requesting microbiome testing without a specific clinical recommendation, because the results don’t reliably translate into actionable medical guidance.
Then, in February 2026, a study published in Communications Biology made the problem even more concrete. Researchers from the University of Maryland and NIST sent three identical stool samples—same exact material—to seven different DTC testing companies. The results that came back were wildly inconsistent. The number of bacterial species identified ranged from a few dozen to several hundred. In some cases, the same sample was flagged as “healthy” by one company and “unhealthy” by another.
Let that sink in. The variability between testing companies was on the same scale as the biological variability between different people. That’s not a minor calibration issue. That’s a fundamental reliability problem.
As one of the study’s co-authors put it: the hype around these products doesn’t match the evidence. And clinicians frequently can’t interpret the results for their patients because there’s no established standard for what a “normal” or “healthy” microbiome even looks like.
Why the Science Isn’t Ready Yet
This isn’t a knock on microbiome science—the field is moving fast and the research is genuinely exciting. But the gap between what we know about the microbiome in general and what we can tell you about your microbiome from a single stool test is enormous. A few key reasons:
There’s no established “healthy reference range.” Unlike a cholesterol level or an HbA1c, we don’t have validated reference ranges for microbiome composition. Your microbiome is shaped by your genetics, diet, geography, medications, age, sleep patterns, stress levels, and dozens of other variables. The inter-individual variability is staggering. So when a test report tells you that your Akkermansia is “low,” it’s comparing you against a database that may or may not reflect people who are actually like you.
Methodological variability is massive. Different companies use different collection methods, lab protocols, sequencing technologies, and data analysis pipelines. Some use 16S rRNA sequencing (identifying bacteria by genus). Some use shotgun metagenomics (more detail, species-level). Viome uses metatranscriptomics (what genes are actively expressed). These are fundamentally different approaches, and they produce fundamentally different reports. There’s no standardization.
Your microbiome is a moving target. Your gut composition changes day to day based on what you ate last night, how you slept, whether you exercised, and even your stress levels. A single snapshot from one stool sample captures a moment in time—not a stable picture of your gut health.
Correlation is still way ahead of causation. We know certain microbial patterns are associated with disease states. But we’re still largely in the descriptive phase of this science. The 2025 Lancet consensus explicitly stated that the field needs to shift from descriptive to mechanistic approaches before these tests have real clinical utility.
So What Can You Actually Do?
Here’s the good news, and honestly it’s the whole reason I wanted to write this post: the evidence base for improving your gut microbiome through lifestyle interventions is much stronger than the evidence for testing it. You don’t need a $300 stool test to start doing the things that actually matter. Here’s what the data supports.
1. Eat More Fermented Foods
This is probably the single strongest lifestyle intervention we have right now for gut diversity, and it comes from a landmark 2021 Stanford study published in Cell by Justin and Erica Sonnenburg’s group.
They randomized 36 healthy adults to either a high-fermented-food diet or a high-fiber diet for 10 weeks. The fermented food group ate 6 servings per day of things like yogurt, kefir, kimchi, sauerkraut, kombucha, and fermented cottage cheese. The results were remarkable: the fermented food group showed a steady, significant increase in overall microbial diversity. Levels of 19 inflammatory proteins dropped, including IL-6, a major driver of chronic inflammation. Four types of immune cells showed less activation.
What was really interesting is that the high-fiber group—despite increasing intake from about 22 to 45 grams per day—did not see a cohort-wide increase in microbial diversity over that short period. The researchers speculated that the microbiome of people eating typical Western diets may be so depleted of fiber-degrading bacteria that it takes longer for fiber to reshape things. Fermented foods, on the other hand, appear to work faster—potentially by directly introducing new microbial species and metabolites into the gut.
Practical takeaway: aim for 2–4 servings of fermented foods daily. Yogurt with live cultures, kefir, kimchi, sauerkraut, miso, and kombucha all count. Go for variety.
2. Dramatically Increase Dietary Fiber (Especially Diversity)
I know I just said the Stanford study didn’t show a short-term diversity bump from fiber. But don’t misread that. The longer-term evidence for dietary fiber and gut health is overwhelming.
A large meta-analysis pooling data from 21 fiber intervention trials and over 2,500 fecal samples found that short-term increases in fiber consumption produced highly consistent gut bacterial responses across studies. Specific clades of bacteria reliably responded to fiber intake. And a 2025 review in Frontiers in Nutrition confirmed that even modest dietary changes can beneficially shift microbial composition and function, particularly by increasing populations of short-chain fatty acid (SCFA) producers like Faecalibacterium prausnitzii and Roseburia.
Why SCFAs matter: butyrate, the most studied SCFA, is the primary fuel for the cells lining your colon. It maintains the intestinal barrier, reduces inflammation, and may play a role in protecting against colorectal cancer. When your fiber intake is low, butyrate production drops, and that appears to directly impair gut barrier function.
The key insight here is diversity of fiber sources, not just total grams. Different fibers feed different bacteria. A tablespoon of psyllium husk isn’t doing the same thing as a serving of lentils, which isn’t doing the same thing as a handful of almonds. You want variety. Legumes, whole grains, nuts, seeds, vegetables, fruits—the wider the range, the more microbial niches you’re feeding.
Practical takeaway: most Americans get about 15 grams of fiber per day. Aim for 30–40 grams from as many different plant sources as possible. Ramp up gradually to avoid GI distress.
3. Exercise Regularly
Exercise independently modifies your gut microbiome in favorable directions. Multiple studies have shown that regular physical activity increases microbial diversity and enriches populations of butyrate-producing bacteria. Athletes consistently demonstrate higher gut diversity than sedentary controls.
The mechanisms likely include improved gut motility, enhanced intestinal blood flow, reduced systemic inflammation, and favorable hormonal signaling. There’s also emerging evidence that exercise modulates the HPA axis (your stress response system) in ways that benefit the gut microbiome—a kind of two-for-one deal.
You don’t need to be an elite athlete. Moderate-intensity exercise—brisk walking, cycling, swimming, resistance training—done consistently appears to be sufficient.
Practical takeaway: 150+ minutes per week of moderate activity. This is already what we recommend for cardiometabolic health, and the microbiome benefits are a bonus.
4. Protect Your Sleep
This one gets less attention than diet and exercise, but the data is compelling. Sleep deprivation has been shown to reduce gut microbial community richness and may damage the intestinal barrier, allowing bacterial endotoxins to leak into circulation and trigger inflammation. The relationship goes both ways: higher gut microbiome diversity correlates with better sleep efficiency and less sleep fragmentation.
The gut microbiota also directly participates in producing neurotransmitters that regulate sleep, including serotonin (the precursor to melatonin) and GABA. Chronic stress, poor sleep, and gut dysbiosis form a vicious cycle mediated through the HPA axis and the vagus nerve.
Practical takeaway: prioritize 7–9 hours of quality sleep. Consistent sleep and wake times matter. This isn’t just “wellness advice”—it has measurable effects on your microbial ecosystem.
5. Cut Ultra-Processed Foods
Western diets high in processed foods, refined sugars, and saturated fats are consistently associated with reduced microbial diversity and expansion of pro-inflammatory bacteria. Clinical data shows that these dietary patterns deplete SCFA-producing species like F. prausnitzii and can elevate circulating TMAO levels (a gut microbiome-derived metabolite linked to cardiovascular disease — check out the great Dr Murphy’s excellent article for more on TMAO) substantially. The effects aren’t subtle—food exchange studies have demonstrated measurable changes in microbial composition and cancer risk markers within just two weeks of switching to a high-processed diet.
Practical takeaway: this doesn’t mean you need to be perfect. But the more you can shift the balance toward whole foods—especially plants—the more you’re feeding the bacteria that keep you healthy.
6. Be Judicious With Antibiotics
This is aimed especially at my physician colleagues. Every unnecessary course of antibiotics is a carpet-bombing of the gut ecosystem. Broad-spectrum antibiotics can obliterate microbial diversity, and recovery can take months to years—if it fully recovers at all. Obviously, use antibiotics when they’re indicated. But the reflexive prescribing of a Z-pack for a viral URI is, from a microbiome perspective, a real cost.
Practical takeaway: if you’re a patient, have an honest conversation with your doctor about whether an antibiotic is truly necessary. If you’re a physician, think about the microbiome cost of that prescription.
7. If You Have Symptoms, Consider a Targeted Elimination Trial
This one needs a caveat up front, because there’s a lot of bad advice floating around on this topic. You’ve probably seen influencers telling everyone to cut gluten and dairy to “reduce inflammation.” The problem is that for people without celiac disease, non-celiac gluten sensitivity, or a documented dairy intolerance, the evidence for blanket elimination is weak—and may actually backfire.
Here’s why. Gluten-free diets in healthy adults have been shown to reduce populations of beneficial bacteria, including Bifidobacterium and Lactobacillus, while also decreasing butyrate production—the exact opposite of what you’re trying to achieve. A randomized crossover trial in 60 healthy Danish adults found that the improvements people reported on a low-gluten diet were likely driven by changes in fiber content, not by removing gluten itself. When you cut gluten-containing grains, you often inadvertently cut a major source of fermentable fiber—whole wheat, barley, rye—that your SCFA-producing bacteria depend on.
As for dairy, it gets even more nuanced. Fermented dairy products—yogurt, kefir, aged cheese—are among the most consistently beneficial foods for the microbiome in the literature. They were literally the star of the Stanford trial I cited earlier. A 2025 narrative review in Nutrition Reviews confirmed that fermented dairy was positively associated with improved GI symptoms, faster colonic transit, and favorable shifts in microbial composition across multiple RCTs. Eliminating all dairy means eliminating one of the best tools we have.
That said—and this is the important part—if you’re dealing with persistent GI symptoms like bloating, irregular bowel habits, skin issues, brain fog, or joint pain, a structured elimination-reintroduction protocol can be genuinely informative. The key word is structured. Remove a suspected trigger (gluten, conventional dairy, or both) for 3–4 weeks, then reintroduce it deliberately and track your symptoms. This is how you generate individualized data about your body—which is more useful than any DTC stool test will give you right now.
For dairy specifically, you can also try swapping rather than eliminating. If conventional milk bothers you, try fermented dairy (kefir, yogurt) or A2 milk before cutting the entire category. Many people who feel lousy after a glass of milk do perfectly well with yogurt or aged cheese, because fermentation breaks down much of the lactose and modifies the protein structure.
Practical takeaway: don’t eliminate gluten or dairy preemptively if you’re asymptomatic. If you do have symptoms, run a proper 3–4 week elimination with systematic reintroduction—ideally with physician guidance. And if you cut gluten-containing grains, make sure you’re replacing the lost fiber from other sources.
The Bottom Line
Gut microbiome science is real, it’s important, and it’s moving fast. The 2025 Gut Microbiota for Health World Summit highlighted advances in microbiome-informed diagnostics, personalized nutrition, and therapeutic innovation. We’re getting closer. But we’re not there yet when it comes to consumer testing.
Right now, the most impactful moves you can make for your microbiome don’t require a test. They require a trip to the grocery store. They require consistent movement. They require decent sleep. And they’re backed by far more robust evidence than anything you’ll get from a DTC stool kit.
Save your $300. Spend it on kimchi, a good pair of walking shoes, and some lentils.
Your gut will thank you.
THE FREE GUT HEALTH PROTOCOL
✔ 2–4 servings of fermented foods daily (yogurt, kefir, kimchi, sauerkraut, kombucha, miso)
✔ 30–40g fiber daily from diverse sources (legumes, whole grains, nuts, seeds, vegetables, fruits)
✔ 150+ minutes moderate exercise per week
✔ 7–9 hours quality sleep with consistent timing
✔ Minimize ultra-processed foods
✔ Avoid unnecessary antibiotics
✔ If symptomatic: structured elimination trial (3–4 weeks elimination, then reintroduce and track)
PS - if you are going to do a gut test I recommend Jona (no affiliation). They have a robust, science/evidence backed platform and are trying to do things the right way. I’ve spoken with their whole team and they have some top minds working there and are putting out solid data. SIBO testing is a caveat where getting the data can help you not only with diagnostics but also therapeautics.
Key References
Porcari S, et al. International consensus statement on microbiome testing in clinical practice. Lancet Gastroenterol Hepatol. 2025;10(2):154-167.
Evaluating the analytical performance of direct-to-consumer gut microbiome testing services. Communications Biology. 2026;9:269.
Wastyk HC, et al. Gut-microbiota-targeted diets modulate human immune status. Cell. 2021;184(16):4137-4153.
Meiners et al. Gut microbiome-mediated health effects of fiber and polyphenol-rich dietary interventions. Front Nutr. 2025.
Short-term dietary fiber interventions produce consistent gut microbiome responses across studies. mSystems. 2024.
Hansen LBS, et al. A low-gluten diet induces changes in the intestinal microbiome of healthy Danish adults. Nat Commun. 2018;9:4630.
De Palma G, et al. Effects of a gluten-free diet on gut microbiota and immune function in healthy adult human subjects. Br J Nutr. 2009;102(8):1154-1160.
Bui G, Marco ML. Impact of fermented dairy on gastrointestinal health and associated biomarkers. Nutr Rev. 2025;nuaf114.
Medical Disclaimer: This post is for educational and informational purposes only and does not constitute medical advice. The content discusses emerging research and off-label considerations in longevity medicine. Always consult your own physician before making changes to your diet, supplement regimen, or medical care. Dr. Sorrell is a practicing longevity medicine physician. This content reflects his clinical perspective informed by the published literature and should not replace individualized medical guidance.